Endoscope image resolution benchmark numbers can be misleading
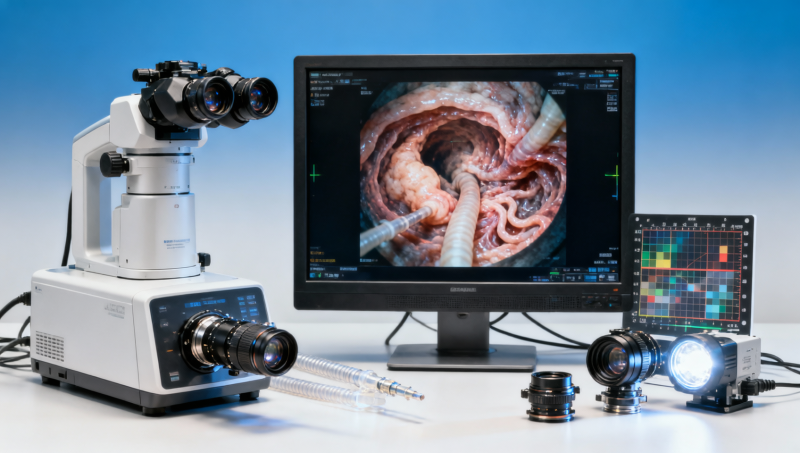
When evaluating an endoscope image resolution benchmark, headline numbers alone can distort real clinical performance. For procurement teams, operators, and decision-makers comparing wholesale medical endoscopes china options, the true picture also depends on medical equipment standards, healthcare compliance, MDR certification, and practical medical device assessment. This article explains why benchmark claims can be misleading and how to verify imaging quality with engineering-grade evidence.
In real purchasing cycles, image quality is rarely determined by a single resolution claim such as 1080p, 2MP, or 4K. Clinical usability depends on how optics, sensor sensitivity, illumination, signal processing, display chain, sterilization durability, and documentation workflow perform together under repeatable conditions. A benchmark sheet may look impressive, yet still fail to reflect what clinicians actually see during a 20-minute procedure.
For hospital procurement directors, MedTech founders, laboratory planners, and daily operators, the risk is practical: selecting an endoscope that scores well in a marketing benchmark but underperforms in low-light anatomy, narrow cavities, or repeated reprocessing cycles. That gap between specification and field performance is exactly where independent technical validation becomes essential.
Why resolution numbers alone do not define clinical image quality
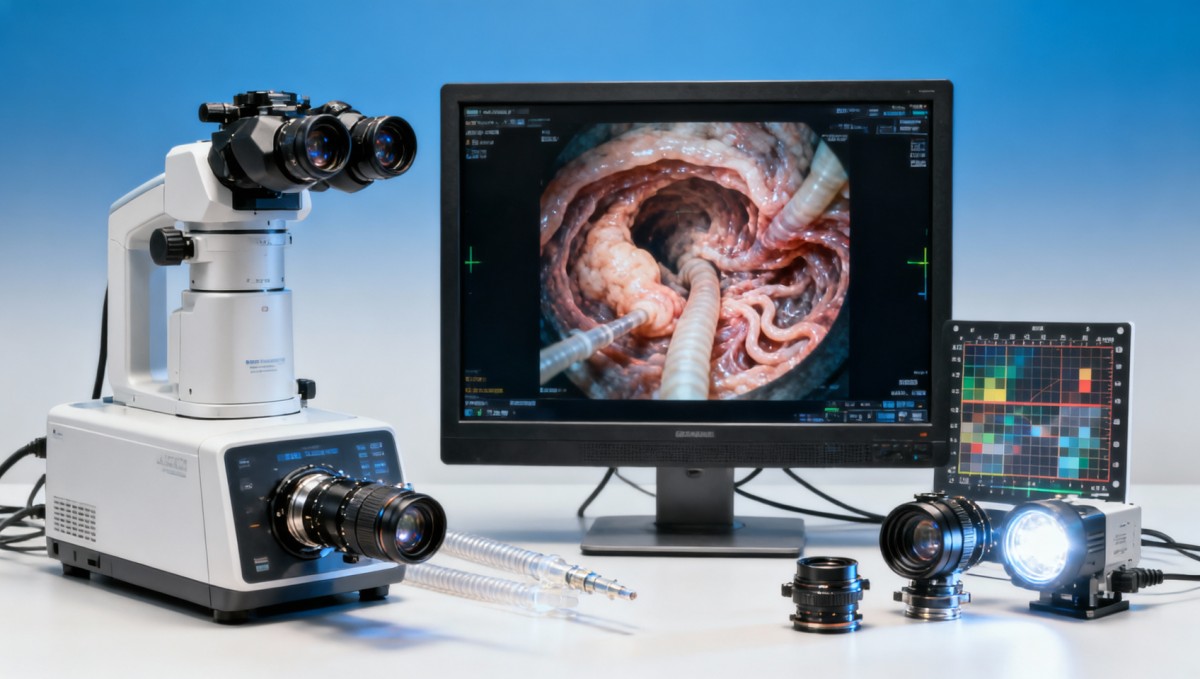
Resolution is only one variable in a much larger imaging system. Two endoscopes with the same stated output, for example 1920 × 1080, can produce visibly different clinical images because pixel count does not describe contrast transfer, edge fidelity, color stability, or signal noise. In practice, one device may preserve mucosal detail at 5–10 mm working distance, while another loses structure under glare or motion.
A common issue is that vendors present sensor resolution rather than effective end-to-end imaging resolution. If the optical path, processor, cable transmission, and monitor scaling each introduce loss, the final displayed image may fall well below the headline number. This is especially relevant when evaluating wholesale medical endoscopes china suppliers, where configuration variation between batches or private-label versions can affect consistency.
Another distortion comes from test setup. A benchmark captured at ideal illumination, fixed distance, and perfectly centered focus may not represent real operating conditions. In many procedures, the field contains fluids, specular highlights, smoke, blood, or rapid hand movement. Under those conditions, noise control, dynamic range, and white balance response can matter as much as nominal sharpness.
For healthcare compliance and practical medical device assessment, buyers should also ask whether a benchmark was measured on the raw sensor, the processed image, or the displayed output. A 3-stage validation path is more reliable: optical bench test, simulated clinical scene test, and in-use workflow review. Without those 3 layers, a resolution benchmark can easily overstate performance.
What resolution fails to capture
- Low-light sensitivity at illumination levels where small vessels or tissue borders become difficult to distinguish.
- Signal-to-noise ratio, especially when gain rises during deep-cavity or narrow-lumen observation.
- Color rendering stability after repeated sterilization or disinfection cycles, often 100–500 cycles depending on device class and use model.
- Latency and motion handling, which affect hand-eye coordination during dynamic procedures.
Typical benchmark mismatch points
In many tenders, four mismatch points appear repeatedly: test distance differs from clinical distance, center sharpness is reported without edge sharpness, image enhancement is activated without disclosure, and display hardware is unspecified. Any one of these can shift perceived quality by a clinically meaningful margin. For precision work, even a 10%–15% loss in edge detail can affect operator confidence.
This is why independent benchmarking laboratories increasingly treat resolution as one metric within a broader quality matrix rather than a standalone purchasing criterion. That broader approach aligns better with value-based procurement and long-term reliability planning.
The technical factors that matter more than a single benchmark score
A clinically useful endoscope imaging system should be assessed as a chain, not a label. The chain usually includes lens design, illumination output, image sensor, processor, transmission path, display device, recording compression, and cleaning-related durability. If one link is weak, the final image can degrade even when the marketed resolution looks competitive.
From an engineering perspective, procurement teams should prioritize at least 6 measurable areas: effective resolution, signal-to-noise ratio, dynamic range, distortion control, color accuracy, and consistency after reprocessing. Operators may add 2 more practical dimensions: anti-fog stability and motion responsiveness. These metrics are more relevant than a single top-line benchmark because they describe how the image behaves during real procedures.
For buyers comparing lower-cost and mid-range systems, a useful rule is to verify performance across 3 working distances and 3 illumination levels. A model that looks strong at one fixed setting may show significant loss when moved from near focus to mid-field observation. The effect is common in rigid and flexible systems alike.
The table below summarizes the difference between headline metrics and decision-grade evaluation criteria used in more rigorous medical device assessment.
The key conclusion is straightforward: a procurement decision based only on resolution can miss failure modes that appear during real use. In most clinical and laboratory environments, stable imaging across time matters more than one impressive number printed in a catalog.
H4-level checkpoints for deeper review
Optics and field uniformity
Ask whether modulation performance drops sharply toward the edge of the field. Uniformity matters when lesions, ports, or instrument tips move away from the center. A field that is sharp in the center but soft in the outer 20%–30% may still produce a strong headline benchmark.
Noise behavior under gain
Low-light compensation can create a cleaner-looking benchmark image while suppressing fine detail. Buyers should request examples with and without aggressive noise reduction to understand the actual trade-off.
How to verify endoscope imaging claims before procurement
A robust verification process does not need to be excessively complex, but it should be structured. For most hospitals, OEM buyers, and enterprise evaluators, a 5-step review process can significantly reduce selection risk. The process should combine documentation review, hands-on testing, standards screening, reprocessing evaluation, and supplier quality checks.
Documentation review should include technical datasheets, test conditions, compatibility notes, service intervals, and any declared medical equipment standards. If MDR certification or related regional compliance is part of the purchasing requirement, the exact scope should be checked carefully. A device family may carry compliance for one configuration while accessories, monitors, or software modules differ by market.
Hands-on testing should simulate real use rather than showroom conditions. This means testing with wet surfaces, variable distances, multiple light settings, and operator movement. A 15–30 minute demo is usually not enough. A more informative evaluation often requires at least 2 use sessions, different operators, and a documented scoring sheet.
The following implementation table can help procurement teams standardize internal review and compare suppliers more fairly.
Once these steps are completed, the team can compare not just image sharpness but total operational suitability. This is particularly important in value-based procurement, where total cost of ownership over 3–5 years may outweigh a lower initial purchase price.
Recommended procurement checklist
- Confirm whether the stated benchmark reflects raw sensor data, processed output, or displayed image.
- Test performance at near, mid, and extended working distance rather than one fixed point.
- Check image behavior with reflections, fluids, and partial occlusion.
- Review reprocessing compatibility and expected service interval, often every 6–12 months depending on use intensity.
- Verify documentation for healthcare compliance, market access scope, and after-sales response commitments.
Compliance, MDR certification, and lifecycle reliability in the real decision process
Imaging performance should never be separated from compliance and durability. An endoscope can produce acceptable images during a short demo but still introduce long-term risk if sealing integrity, cable strain relief, connector stability, or validated cleaning compatibility are weak. In regulated healthcare procurement, those issues can affect uptime, traceability, and replacement planning.
For cross-border sourcing, including wholesale medical endoscopes china evaluation, buyers should review how technical claims align with regional regulatory pathways. MDR certification discussions often focus on legal market access, but operational teams also need supporting evidence on labeling consistency, intended-use documentation, and change control. A product revision that appears minor may still influence image processing behavior or accessory compatibility.
Lifecycle reliability also matters because image degradation may be gradual. A device can pass initial acceptance and still show declining brightness, focus stability, or connector integrity after repeated cleaning cycles. For high-turnover environments, even a modest drop in image consistency over 6–12 months can affect confidence and maintenance cost. Procurement teams should therefore request evidence of durability testing, not only day-one imaging samples.
Independent technical benchmarking adds value here by translating scattered supplier claims into comparable engineering evidence. Instead of asking which product has the highest advertised resolution, decision-makers can ask a more useful question: which product maintains clinically acceptable imaging, compliance alignment, and serviceability across the intended use life?
Risk areas often missed in tenders
- Configuration drift between demo units and delivered production lots.
- Incomplete disclosure of monitor, processor, or compression settings used in benchmark demonstrations.
- Unclear maintenance intervals, spare parts lead time, or repair turnaround that may exceed 2–6 weeks.
- Insufficient traceability of software or firmware changes that alter image enhancement behavior.
What enterprise decision-makers should request
A solid supplier package should include a controlled test summary, declared operating conditions, reprocessing guidance, service pathway, and change notification process. These are not administrative extras. They are decision-critical inputs when comparing technical integrity and long-term reliability across multiple vendors.
FAQ: practical questions from researchers, users, buyers, and executives
How should I compare two endoscopes that both claim 4K or HD output?
Start with like-for-like testing. Use the same monitor, same illumination conditions, same working distance, and the same recording path. Then compare center and edge detail, low-light noise, color stability, and latency. If possible, score performance across at least 5 criteria rather than relying on one image sample.
Are lower-cost sourced products always risky?
Not necessarily. Cost alone does not determine quality. The real issue is evidence depth. Some suppliers provide clear technical files, repeatable test conditions, and stable manufacturing controls, while others do not. A disciplined medical device assessment can separate economically attractive options from technically uncertain ones.
What is a reasonable evaluation period before purchase approval?
For standard procurement, 2–4 weeks is often a realistic period to complete document review, controlled testing, and workflow simulation. Complex enterprise evaluations may take longer if multiple departments, compliance reviews, or reprocessing teams need to sign off.
Which metrics matter most for operators?
Operators usually care most about usable detail, stable color, low glare, predictable focus, and minimal lag. These factors directly affect visibility and handling confidence during live procedures. A technically high benchmark means little if the image becomes unstable once motion, fluids, or reflections enter the field.
Endoscope image resolution benchmark numbers can be useful, but only when placed in the right context. For serious healthcare procurement, the better question is not who advertises the highest number, but who can prove effective image quality, compliance alignment, and lifecycle reliability under realistic conditions. That is where independent benchmarking, structured evaluation, and engineering-grade interpretation create real purchasing confidence.
VitalSync Metrics supports data-driven medical technology decisions by turning technical claims into comparable evidence for procurement directors, operators, MedTech teams, and laboratory architects. If you need a deeper benchmark review, supplier comparison framework, or custom medical device assessment workflow, contact us today to discuss your application and get a tailored evaluation plan.
- EMS
- ESS
- PPE
- EdTech
- procurement
- AR
- Cement
- benchmarking
- benchmarking lab
- healthcare procurement
- MDR certification
- value-based procurement
- medical device assessment
- medical equipment standards
- medical technology
- technical integrity
- medical equipment
- hospital procurement
- technical benchmarking
- signal-to-noise ratio
- long-term reliability
- data-driven
- clinical performance
- healthcare compliance
- wholesale medical endoscopes china
- endoscope image resolution benchmark
Recommended News
- 2026.04.23Low-Altitude Economy Enters Scale-Up Year: eVTOL Infrastructure & Vital Sign SensingLydia Vancini (Regulatory Compliance Lead)
- 2026.04.23Lianyirong, ABC Shanghai Dongfang Hub Branch Close Two e-NI TransactionsDr. Hideo Tanaka (Imaging Systems Analyst)
- 2026.04.23China’s April 2026 LPR Set at 3.0% (1Y) and 3.5% (5Y+): Implications for Medical Device Export Financing
The VitalSync Intelligence Brief
Receive daily deep-dives into MedTech innovations and regulatory shifts.
