lndustry Portal
Global B2B Solutions
 ISO 13485:2025 Draft Tightens Traceability to Raw MaterialsISO 13485:2025 draft mandates raw material traceability—key for medtech suppliers, OEMs & regulators. Act now to align QMS before Q1 2027 launch.May 30, 2026
ISO 13485:2025 Draft Tightens Traceability to Raw MaterialsISO 13485:2025 draft mandates raw material traceability—key for medtech suppliers, OEMs & regulators. Act now to align QMS before Q1 2027 launch.May 30, 2026 FDA Tightens 510(k) Review for AI in Remote Monitoring DevicesFDA tightens 510(k) review for AI in remote monitoring devices—learn how new clinical validation rules impact manufacturers, suppliers & compliance strategies.May 30, 2026
FDA Tightens 510(k) Review for AI in Remote Monitoring DevicesFDA tightens 510(k) review for AI in remote monitoring devices—learn how new clinical validation rules impact manufacturers, suppliers & compliance strategies.May 30, 2026 EUDAMED Core Modules Go Mandatory May 28, 2026; UDI Registration Required for EU Customs ClearanceEUDAMED core modules go mandatory May 28, 2026—UDI registration & SRN now essential for EU customs clearance. Act now to avoid delays at Rotterdam, Hamburg & beyond.May 30, 2026
EUDAMED Core Modules Go Mandatory May 28, 2026; UDI Registration Required for EU Customs ClearanceEUDAMED core modules go mandatory May 28, 2026—UDI registration & SRN now essential for EU customs clearance. Act now to avoid delays at Rotterdam, Hamburg & beyond.May 30, 2026 Are playground safety standards en1176 still enough?Playground safety standards en1176 remain essential—but are they enough? Discover data-led inspection strategies to reduce risk and protect every play environment.May 30, 2026Dr. Alistair Thorne (Senior Biomedical Engineer)
Are playground safety standards en1176 still enough?Playground safety standards en1176 remain essential—but are they enough? Discover data-led inspection strategies to reduce risk and protect every play environment.May 30, 2026Dr. Alistair Thorne (Senior Biomedical Engineer) Why does a Medical diagnostic equipment price list vary?Medical diagnostic equipment price list varies by compliance, software, service, consumables, and lifecycle risk. Learn how to compare quotes and buy with confidence.May 30, 2026Dr. Hideo Tanaka (Imaging Systems Analyst)
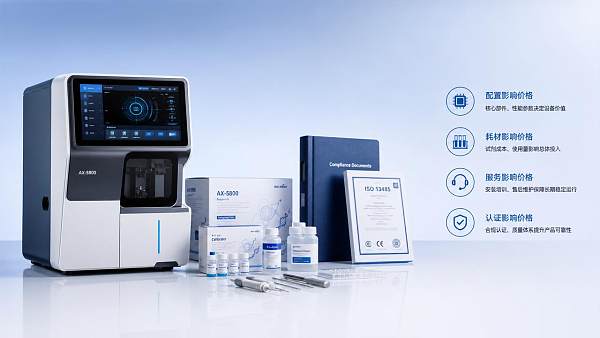
Why does a Medical diagnostic equipment price list vary?Medical diagnostic equipment price list varies by compliance, software, service, consumables, and lifecycle risk. Learn how to compare quotes and buy with confidence.May 30, 2026Dr. Hideo Tanaka (Imaging Systems Analyst) Where is healthcare technology cutting real costs?Healthcare technology can cut real costs when evidence proves reliability, workflow fit, and lifecycle value. See how to separate true savings from costly promises.May 30, 2026Lydia Vancini (Regulatory Compliance Lead)
Where is healthcare technology cutting real costs?Healthcare technology can cut real costs when evidence proves reliability, workflow fit, and lifecycle value. See how to separate true savings from costly promises.May 30, 2026Lydia Vancini (Regulatory Compliance Lead) What makes a car batteries supplier worth trusting?car batteries supplier selection made smarter: learn how to verify quality data, compliance, testing standards, warranty risk, and long-term supply reliability before you buy.May 30, 2026Dr. Alistair Thorne (Senior Biomedical Engineer)
What makes a car batteries supplier worth trusting?car batteries supplier selection made smarter: learn how to verify quality data, compliance, testing standards, warranty risk, and long-term supply reliability before you buy.May 30, 2026Dr. Alistair Thorne (Senior Biomedical Engineer) How to choose manufacturing components without reworkManufacturing components selection made smarter: learn how to verify specs, suppliers, materials, and lifecycle risks to avoid costly rework and improve production confidence.May 30, 2026Dr. Julian Rossi (RehabTech Specialist)
How to choose manufacturing components without reworkManufacturing components selection made smarter: learn how to verify specs, suppliers, materials, and lifecycle risks to avoid costly rework and improve production confidence.May 30, 2026Dr. Julian Rossi (RehabTech Specialist) NMPA Cracks Down on 4 Illegal Online Sales of Remote ECG & Home Vital MonitorsNMPA cracks down on illegal online sales of remote ECG & home vital monitors—key alert for cross-border sellers, e-commerce platforms, and distributors. Act now to ensure NMPA filing compliance.May 29, 2026
NMPA Cracks Down on 4 Illegal Online Sales of Remote ECG & Home Vital MonitorsNMPA cracks down on illegal online sales of remote ECG & home vital monitors—key alert for cross-border sellers, e-commerce platforms, and distributors. Act now to ensure NMPA filing compliance.May 29, 2026
 {item.title}{item.description}{item.publish_time}
{item.title}{item.description}{item.publish_time}Search News
Popular Tags
Industry Overview
The global commercial kitchen equipment market is projected to reach $112 billion by 2027. Driven by urbanization, the rise of e-commerce food delivery, and strict hygiene regulations.