lndustry Portal
Global B2B Solutions
 Medical device assessment questions before choosing roboticsMedical device assessment questions before choosing robotics: compare medical device evaluation, testing, MDR IVDR readiness, compliance, integration, and reliability to make smarter healthcare procurement decisions.Apr 17, 2026Dr. Alistair Thorne (Senior Biomedical Engineer)
Medical device assessment questions before choosing roboticsMedical device assessment questions before choosing robotics: compare medical device evaluation, testing, MDR IVDR readiness, compliance, integration, and reliability to make smarter healthcare procurement decisions.Apr 17, 2026Dr. Alistair Thorne (Senior Biomedical Engineer) Medical regulatory compliance risks in connected sensor dataMedical regulatory compliance risks in connected sensor data: explore medical device testing, medical device evaluation, MDR IVDR alignment, and practical steps to protect reliability, compliance, and safer adoption.Apr 17, 2026Lydia Vancini (Regulatory Compliance Lead)
Medical regulatory compliance risks in connected sensor dataMedical regulatory compliance risks in connected sensor data: explore medical device testing, medical device evaluation, MDR IVDR alignment, and practical steps to protect reliability, compliance, and safer adoption.Apr 17, 2026Lydia Vancini (Regulatory Compliance Lead) Medical regulatory compliance challenges in device packagingMedical regulatory compliance in device packaging: explore MDR IVDR risks, medical device evaluation, testing, and compliance benchmarks to help global decision-makers source with confidence.Apr 17, 2026Dr. Julian Rossi (RehabTech Specialist)
Medical regulatory compliance challenges in device packagingMedical regulatory compliance in device packaging: explore MDR IVDR risks, medical device evaluation, testing, and compliance benchmarks to help global decision-makers source with confidence.Apr 17, 2026Dr. Julian Rossi (RehabTech Specialist) Which medical equipment standards matter for sample storageMedical equipment standards for sample storage: learn how MDR IVDR, laboratory equipment validation, and medical equipment compliance shape safer, auditable, reliable buying decisions.Apr 17, 2026Dr. Hideo Tanaka (Imaging Systems Analyst)
Which medical equipment standards matter for sample storageMedical equipment standards for sample storage: learn how MDR IVDR, laboratory equipment validation, and medical equipment compliance shape safer, auditable, reliable buying decisions.Apr 17, 2026Dr. Hideo Tanaka (Imaging Systems Analyst) Medical equipment standards that affect ultrasound upgradesMedical equipment standards guide smarter ultrasound upgrades through medical device testing, MDR IVDR review, and medical technology assessment—helping hospitals reduce compliance risk and improve long-term value.Apr 17, 2026Lydia Vancini (Regulatory Compliance Lead)
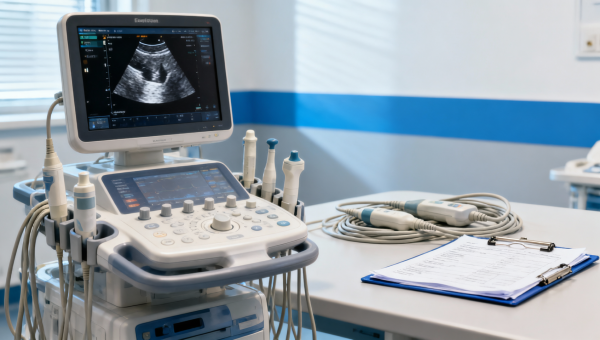
Medical equipment standards that affect ultrasound upgradesMedical equipment standards guide smarter ultrasound upgrades through medical device testing, MDR IVDR review, and medical technology assessment—helping hospitals reduce compliance risk and improve long-term value.Apr 17, 2026Lydia Vancini (Regulatory Compliance Lead) How medical device reliability fails under repeated sterilizationMedical device reliability under repeated sterilization: discover how medical device testing, medical device evaluation, and healthcare benchmarking expose compliance risks before failure.Apr 17, 2026Dr. Alistair Thorne (Senior Biomedical Engineer)
How medical device reliability fails under repeated sterilizationMedical device reliability under repeated sterilization: discover how medical device testing, medical device evaluation, and healthcare benchmarking expose compliance risks before failure.Apr 17, 2026Dr. Alistair Thorne (Senior Biomedical Engineer) Medical device reliability problems in centrifugation techMedical device reliability in centrifugation tech: explore medical device testing, medical device evaluation, healthcare benchmarking, and MDR IVDR-ready compliance insights for smarter procurement.Apr 17, 2026Dr. Hideo Tanaka (Imaging Systems Analyst)
Medical device reliability problems in centrifugation techMedical device reliability in centrifugation tech: explore medical device testing, medical device evaluation, healthcare benchmarking, and MDR IVDR-ready compliance insights for smarter procurement.Apr 17, 2026Dr. Hideo Tanaka (Imaging Systems Analyst) Medical technology compliance checks for rehabilitation robotsMedical technology compliance for rehabilitation robots: explore medical device evaluation, testing, MDR IVDR alignment, and benchmarking to reduce procurement risk and buy with confidence.Apr 17, 2026Dr. Alistair Thorne (Senior Biomedical Engineer)
Medical technology compliance checks for rehabilitation robotsMedical technology compliance for rehabilitation robots: explore medical device evaluation, testing, MDR IVDR alignment, and benchmarking to reduce procurement risk and buy with confidence.Apr 17, 2026Dr. Alistair Thorne (Senior Biomedical Engineer) Medical technology compliance risks in connected orthoticsMedical technology compliance risks in connected orthotics: learn how medical device testing, medical technology evaluation, and MDR IVDR checks reduce procurement risk and improve reliability.Apr 17, 2026Sarah Jenkins (Laboratory Infrastructure Consultant)
Medical technology compliance risks in connected orthoticsMedical technology compliance risks in connected orthotics: learn how medical device testing, medical technology evaluation, and MDR IVDR checks reduce procurement risk and improve reliability.Apr 17, 2026Sarah Jenkins (Laboratory Infrastructure Consultant)
 {item.title}{item.description}{item.publish_time}{item.goods_category_sif3qfva9d}
{item.title}{item.description}{item.publish_time}{item.goods_category_sif3qfva9d}Search News
Popular Tags
Industry Overview
The global commercial kitchen equipment market is projected to reach $112 billion by 2027. Driven by urbanization, the rise of e-commerce food delivery, and strict hygiene regulations.