Choosing ultrasound transducer frequency range for shallow structures
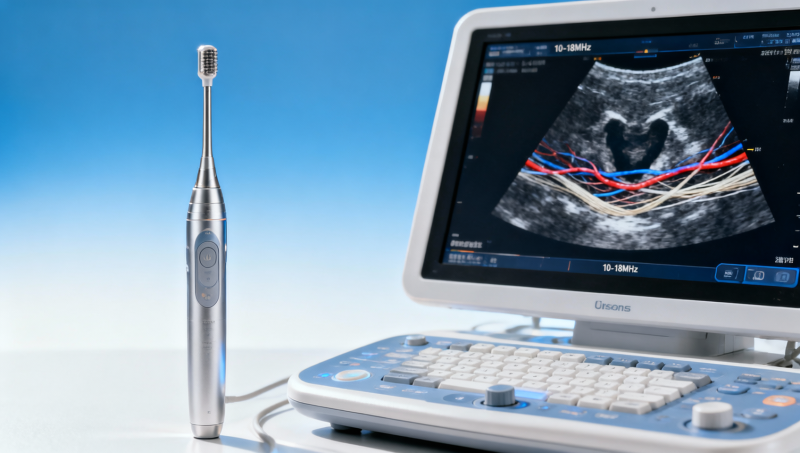
Selecting the right ultrasound transducer frequency range for shallow structures is critical to achieving precise imaging without sacrificing penetration or workflow efficiency. For clinicians, buyers, and MedTech evaluators, this decision also connects with broader quality concerns such as signal to noise ratio in patient monitors, biocompatibility testing standards, and ISO 13485 audit requirements. This guide explains how to assess frequency choices with technical clarity and procurement confidence.
In practical terms, frequency selection affects image sharpness, lesion conspicuity, exam speed, operator confidence, and downstream purchasing decisions. A transducer that performs well in superficial vascular access, thyroid scans, musculoskeletal imaging, or small-parts assessment must balance axial resolution, lateral resolution, and sufficient penetration within a limited depth window, often between 1 cm and 6 cm.
For hospital procurement teams and MedTech decision-makers, this is not only a clinical question. It also involves lifecycle cost, probe durability, compatibility with installed ultrasound platforms, cleaning validation, and evidence that engineering claims match measurable output. That is why frequency range should be reviewed as part of a broader technical benchmarking process rather than as a marketing specification in isolation.
Why Frequency Range Matters for Shallow Structure Imaging
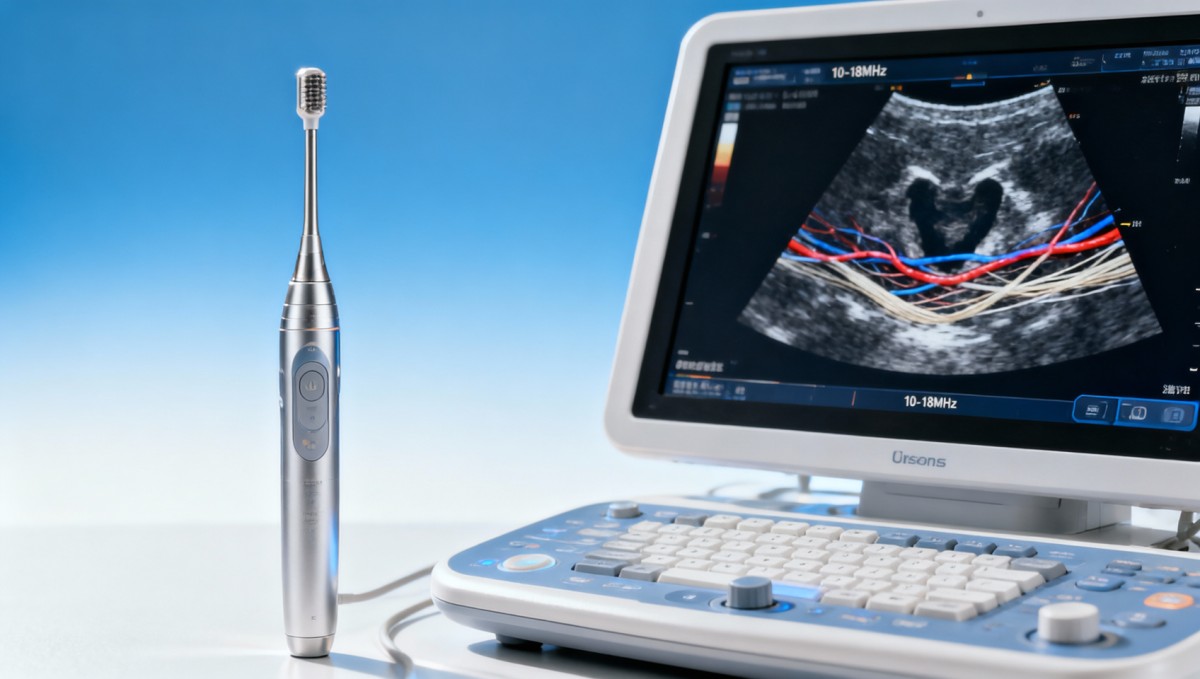
Ultrasound frequency determines the tradeoff between resolution and penetration. Higher frequencies, typically in the 10–18 MHz range, produce shorter wavelengths and therefore finer detail. This is highly valuable when imaging superficial tendons, nerves, breast lesions, finger joints, pediatric soft tissue, or vascular structures located within the first 2–4 cm beneath the skin surface.
However, no probe should be selected on peak frequency alone. A broad usable bandwidth matters because superficial exams vary. For example, skin and subcutaneous targets may require 15–18 MHz for edge definition, while deeper small-parts views may need 7–12 MHz to preserve adequate penetration and reduce dropout. A narrow-band probe can force repeated adjustments and reduce workflow efficiency in busy departments.
Operators also need to consider how frequency interacts with beamforming, focal zones, and signal processing. Two probes labeled as “high frequency” may produce different real-world images because cable shielding, element sensitivity, backing material, and system software affect signal-to-noise ratio. In procurement reviews, image clarity at 3 cm depth may be more meaningful than headline frequency claims alone.
From a quality and compliance perspective, selecting the correct transducer frequency range reduces rescans, shortens exam time by 10–20% in some routine workflows, and supports more consistent documentation. For B2B buyers, that translates into better utilization of installed systems, lower training burden, and a clearer basis for comparing vendors beyond brochure language.
Typical frequency bands by superficial application
The following table provides a practical mapping between common shallow imaging tasks and the frequency range usually considered effective in routine clinical use. Final selection still depends on patient habitus, probe footprint, and system electronics.
A key takeaway is that “higher” is not always “better.” For many shallow exams, a flexible 8–15 MHz or 10–15 MHz linear transducer offers more practical value than an ultra-high-frequency probe that loses performance beyond 2–3 cm. Procurement teams should therefore ask for depth-specific sample images and not just nominal frequency labels.
How to Match Frequency Range to Clinical Use Cases and Operators
The best ultrasound transducer frequency range depends on who is using the system, what structures are being scanned, and how often the probe changes hands across departments. A radiology team focused on thyroid and breast imaging may prioritize consistency and grayscale detail, while anesthesia or vascular access teams may value needle guidance, rapid boot-up behavior, and easy near-field optimization.
For shallow structures, linear array probes are usually the first choice because they provide a rectangular field of view and high near-field resolution. In many hospitals, a 5–12 MHz or 6–15 MHz probe becomes the general-purpose superficial workhorse. A second probe in the 12–18 MHz class may then be justified for specialized musculoskeletal or dermatologic use where structures lie within the first 20 mm.
Operator skill also matters. Novice users often benefit from a wider bandwidth probe and presets that stabilize image quality across variable scanning angles. Advanced users may exploit higher frequencies more effectively, especially in tendon, nerve, or interventional guidance cases. This means procurement should align with training level, not only with top-end technical capability.
If one platform must serve multiple departments, the selection process should document at least 4 factors: target depth, required resolution, expected exam volume, and sterilization or disinfection workflow. A probe that performs strongly in 70% of superficial tasks may be a better investment than a premium specialty transducer used in only 10% of exams.
A practical selection framework
- Define the target anatomy by actual depth bands such as 0–2 cm, 2–4 cm, and 4–6 cm rather than by department name alone.
- Review whether detail recognition, Doppler sensitivity, or needle visibility is the primary task in at least 3 representative exam types.
- Check system compatibility, footprint size, cable strain resistance, and cleaning protocol before ranking image quality only.
- Run side-by-side trials using identical gain, focus, depth, and preset logic to avoid biased comparisons.
- Document usability feedback from at least 2 user groups, such as sonographers and procedural operators.
When a broader bandwidth is preferable
A broader transducer frequency range is particularly useful in mixed outpatient settings, emergency departments, and procedure rooms where patient body habitus and exam goals change quickly. Instead of switching probes repeatedly, users can step down from 15 MHz to 8 MHz or 7 MHz while maintaining acceptable image continuity and saving several minutes per case.
Technical Benchmarks Procurement Teams Should Request
A sound purchasing decision requires measurable evidence. For superficial imaging probes, buyers should request technical benchmarking that goes beyond frequency labels and demo-room impressions. Useful documentation may include axial and lateral resolution at defined depths, dead zone performance, uniformity across the footprint, Doppler sensitivity in small vessels, and signal-to-noise behavior under standard presets.
In a value-based procurement model, the transducer should also be assessed for durability and compliance. Probe housing materials, lens integrity, and cable relief design influence service life, especially in departments running 20–40 scans per day. Cleaning chemistry compatibility and biocompatibility relevance are also important because superficial probes often have frequent skin contact and repeated reprocessing cycles.
For organizations operating under strict quality systems, vendor responses should support internal review against documented procedures. That includes traceability of test methods, alignment with manufacturing quality controls, and evidence suitable for supplier qualification discussions linked to MDR/IVDR-facing workflows or ISO 13485 audit readiness. The goal is not to overregulate the purchase, but to avoid equipment that looks adequate in a short demonstration and underperforms over 12–36 months.
Independent benchmarking bodies such as VitalSync Metrics are valuable in this process because they translate raw engineering characteristics into standardized review criteria. For decision-makers comparing several suppliers, this narrows the gap between promotional claims and deployable clinical performance.
Core benchmarks to compare before purchase
The table below summarizes the most useful parameters to request during supplier evaluation for shallow-structure transducers.
The strongest suppliers are usually those who can explain how these parameters were measured, what conditions were used, and where limitations exist. A vendor unwilling to provide depth-specific evidence or durability context may create risk later in acceptance testing and long-term ownership.
Common Mistakes in Selecting a Shallow Imaging Transducer
One frequent mistake is assuming that the highest advertised MHz value guarantees the best superficial image. In reality, excessive focus on top-end frequency can lead to inadequate penetration, inconsistent brightness, or poor utility outside a narrow depth band. A 17 MHz probe may look impressive on paper, yet a 12–15 MHz transducer can outperform it in mixed workloads where depth varies between 2 cm and 5 cm.
A second mistake is separating clinical evaluation from procurement evaluation. Departments sometimes approve a probe after a short demonstration without considering repair frequency, transducer lens wear, connector fragility, and cleaning burden. Over a 24-month period, these issues may matter more than minor differences in brochure specifications.
Another common issue is testing only on ideal patients. Shallow imaging still changes with edema, obesity, surgical dressings, or difficult access angles. A useful evaluation should include at least 3 realistic scenarios and more than 1 operator. This reduces the risk of selecting a probe that works well in controlled demos but inconsistently in real workflows.
Finally, some organizations overlook integration. If the transducer frequency range is suitable but presets are poorly optimized, if Doppler sensitivity is unstable, or if the installed ultrasound platform handles beamforming inconsistently, the actual user experience may remain suboptimal. The probe and the host system must be reviewed as a combined imaging chain.
Risk checklist for buyers and evaluators
- Do not compare probes using different presets, gain settings, or focal zones, because this can distort real performance differences.
- Do not ignore cable and connector fatigue if the expected use exceeds 15 scans per day in procedure-heavy departments.
- Do not treat frequency range as the only specification; footprint, ergonomics, Doppler response, and software stability also affect adoption.
- Do not assume compliance documentation is separate from performance; audit-ready supplier records reduce downstream qualification friction.
A balanced decision principle
The most reliable choice is often the transducer that delivers repeatable image quality across 80% of target cases, supports efficient handling, and has verifiable engineering consistency. This is especially true for procurement teams seeking lower total cost of ownership rather than isolated technical extremes.
Implementation, Validation, and Procurement Guidance
Once the preferred ultrasound transducer frequency range has been identified, organizations should move into a structured implementation phase. This usually includes 3 stages: technical verification, user acceptance, and post-installation review. A well-run rollout can reduce training friction and reveal whether the transducer meets both image-quality and operational expectations within the first 30–60 days.
Technical verification should confirm compatibility with existing ultrasound systems, accessory interfaces, cleaning instructions, and data documentation requirements. User acceptance should involve at least 2 departments when the probe serves multiple workflows. Post-installation review should capture service response times, reprocessing ease, and any image consistency concerns reported by frontline operators.
For enterprise buyers, supplier dialogue should include replacement lead time, warranty scope, preventive handling guidance, and availability of technical benchmarking data for future tenders. In many procurement cycles, the difference between a low-risk purchase and a problematic one is the quality of documentation available before signing, not after deployment.
VitalSync Metrics supports this approach by converting engineering details into decision-ready evidence. For hospitals, MedTech startups, and lab planners, that means clearer supplier comparisons, less dependence on promotional language, and stronger confidence that the selected transducer frequency range will perform in routine clinical use.
Suggested implementation workflow
- Define the top 3 to 5 target superficial applications and their depth ranges.
- Request benchmark images and technical documentation from shortlisted suppliers.
- Run controlled user trials over 1–2 weeks with standardized presets.
- Score performance using image quality, usability, durability indicators, and service commitments.
- Approve deployment with a 30-day and 90-day review checkpoint.
FAQ: short answers for common buying questions
How high should frequency be for shallow structures? In many routine superficial exams, 10–15 MHz is a practical starting range. Highly superficial nerves, tendons, and skin-adjacent structures may benefit from 15–18 MHz if penetration remains adequate.
Is a single probe enough for all shallow applications? Sometimes yes, especially with a broad-band linear probe such as 6–15 MHz. But high-volume or specialty centers often pair a general superficial probe with an ultra-high-frequency option for advanced musculoskeletal detail.
What should procurement teams prioritize? Focus on 4 areas: verified image quality at target depths, usable bandwidth, durability under expected scan volume, and documentation quality for supplier qualification and service planning.
Choosing the right ultrasound transducer frequency range for shallow structures requires more than selecting the highest MHz value. The best decision aligns depth needs, operator workflow, measurable image performance, durability, and supplier transparency. When evaluation includes technical benchmarks, realistic use cases, and lifecycle considerations, buyers can reduce risk while improving clinical consistency.
If your team needs support comparing superficial imaging probes, validating supplier claims, or translating engineering data into procurement-ready criteria, VitalSync Metrics can help you build a clearer and more defensible decision path. Contact us to discuss a customized evaluation framework, request technical benchmarking guidance, or explore broader healthcare sourcing solutions.
Recommended News
- 2026.06.02Smart Street Lighting Cost-Effective Solutions: Controls, LEDs, and ROILydia Vancini (Regulatory Compliance Lead)
- 2026.06.02Stepper Motors Selection Guide: Torque, Step Angle, Drivers, and Load FitDr. Alistair Thorne (Senior Biomedical Engineer)
- 2026.06.02UV Water Sterilizers for Small Facilities: Flow Rate, Dose, and Lamp LifeDr. Alistair Thorne (Senior Biomedical Engineer)
The VitalSync Intelligence Brief
Receive daily deep-dives into MedTech innovations and regulatory shifts.
