Diagnostic Imaging software: what to compare first
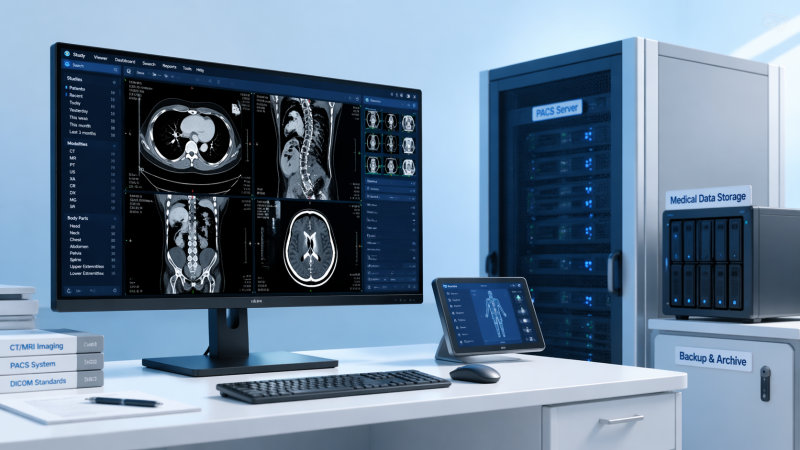
Choosing Diagnostic Imaging software is not just about features—it starts with comparing clinical accuracy, interoperability, regulatory alignment, and long-term medical technology cost. For procurement teams, operators, and healthcare decision-makers, the right evaluation framework reduces risk and supports medical equipment safety standards, medical equipment calibration, and broader healthcare compliance solutions in an increasingly data-driven care environment.
In practice, diagnostic imaging software affects far more than image viewing. It influences reporting speed, workflow consistency, archive strategy, cybersecurity exposure, and even whether a site can scale from 1 modality to 5 or more without a costly platform reset. For hospitals, imaging centers, laboratory planners, and MedTech innovators, the first comparison points should be technical and operational, not cosmetic.
This is where a benchmarking mindset matters. VitalSync Metrics (VSM) focuses on separating promotional claims from measurable performance so buyers and operators can evaluate imaging platforms on evidence: image fidelity, integration depth, validation process, uptime targets, support responsiveness, and lifecycle cost over 3–7 years.
Start with clinical accuracy, image integrity, and diagnostic reliability
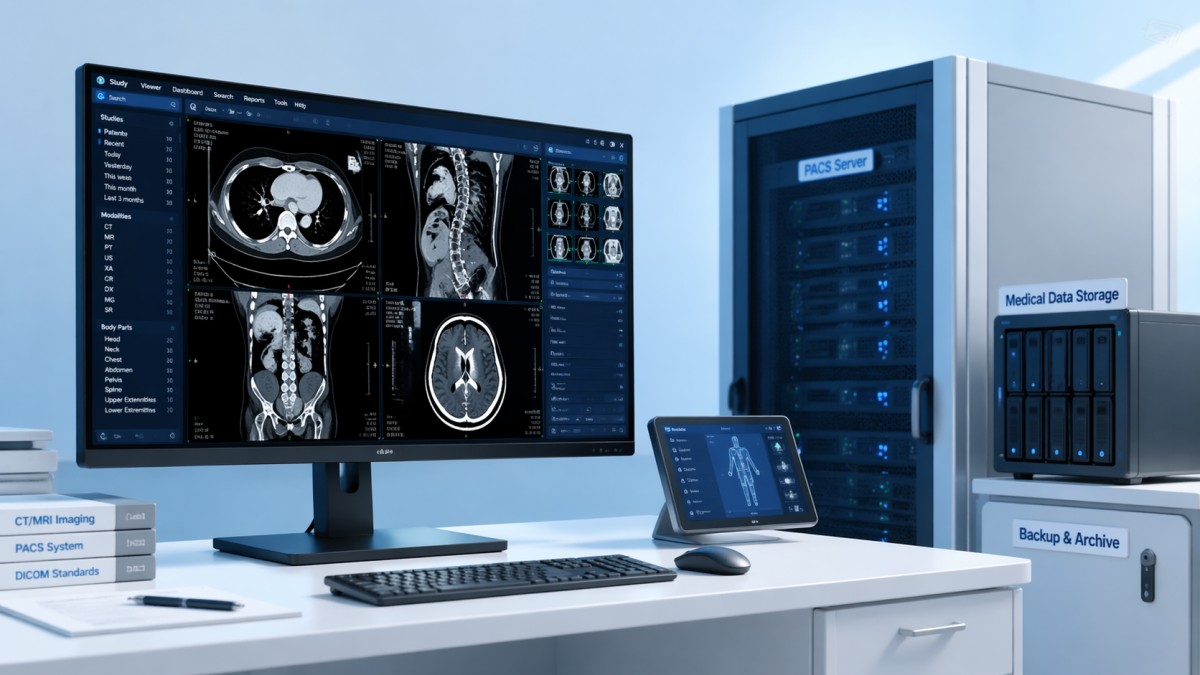
The first comparison for diagnostic imaging software should be whether the system preserves clinical meaning across acquisition, processing, visualization, and reporting. An elegant interface has little value if image rendering alters grayscale interpretation, loses metadata, or introduces workflow shortcuts that increase reading risk. In radiology, cardiology, pathology imaging, and hybrid care settings, even a small display or annotation inconsistency can affect downstream decisions.
Buyers should ask how the platform manages DICOM fidelity, hanging protocols, image compression, and version control. A practical review should include at least 4 checkpoints: native image preservation, annotation traceability, viewer consistency across workstations, and measurable latency under load. For example, if a study opens in 2–4 seconds on one station but takes 10–15 seconds on another, workflow reliability is already compromised.
Operators should also compare the software’s support for calibration-sensitive environments. If diagnostic viewing is performed on calibrated displays, the software should work predictably with medical equipment calibration procedures, grayscale presentation standards, and audit logs. Inconsistent rendering between acquisition room, reading room, and remote review station creates avoidable clinical risk.
From a procurement perspective, it helps to compare performance under realistic volumes. A small center may process 50–100 studies per day, while a multi-site network may handle 1,000 or more. Software that performs well in a demo with 20 sample studies may behave very differently in live conditions with concurrent users, archive queries, and AI-assisted workflows running together.
Core technical questions to test first
- Does the platform preserve original image data and metadata without non-transparent modification during import, export, or compression?
- Can the system maintain acceptable viewer response times below 3 seconds for common studies and below 8 seconds for larger datasets under normal network load?
- Are measurements, annotations, and reporting links traceable across 2 or more user roles such as technologist, radiologist, and referring clinician?
- Does the software support structured quality checks for monitor calibration, user audit trails, and change history over a retention period of 12–36 months?
The table below shows a practical way to compare clinical performance priorities before discussing optional modules or visual design preferences.
A clear takeaway is that clinical accuracy must be tested before convenience features. If image integrity, traceability, and loading performance are not stable, future upgrades or AI tools will only amplify baseline problems rather than solve them.
Compare interoperability before comparing advanced features
The second priority is interoperability. Diagnostic imaging software rarely works alone. It must communicate with RIS, HIS, EHR, PACS, laboratory systems, billing tools, and in many cases cloud archives or vendor-neutral archives. A platform with 20 advanced features but weak integration can create more manual steps, duplicate records, and data reconciliation work than a simpler but better-connected system.
Healthcare teams should compare support for DICOM, HL7, FHIR-aligned workflows where relevant, user directory integration, and API accessibility. In a typical procurement review, at least 5 data exchange questions should be documented: patient identity matching, study routing, report delivery, archive retrieval, and downtime recovery. These are not technical side notes; they directly influence operator efficiency and patient safety.
Interoperability also determines implementation speed. If the software can connect to existing systems with standard mapping and predictable validation, deployment may take 4–8 weeks. If heavy customization is required, timelines can extend to 3–6 months, especially across multi-site organizations with different modality vendors and legacy archives.
For enterprise decision-makers, it is useful to evaluate not only current compatibility but future flexibility. Can the platform support 2 sites today and 10 sites later? Can new modalities, AI triage modules, or teleradiology workflows be added without replacing the archive core? Scalability should be measured in interfaces, users, and study volume, not promised in generic terms.
Interoperability checkpoints that reduce procurement risk
- Validate data standards support, including import, export, and exception handling for common DICOM and HL7 scenarios.
- Review workflow mapping for scheduling, acquisition, interpretation, report finalization, and downstream delivery.
- Confirm whether integrations are standard connectors or custom builds that may increase maintenance cost by 15%–30% over time.
- Ask for test protocol details covering failed transmission, duplicate patient records, and archive restoration.
- Check whether role-based access and audit functions remain intact across integrated systems.
The next table helps compare integration maturity in a structured way during vendor or platform review.
A system with stronger interoperability usually produces hidden savings. Fewer manual corrections, fewer failed study transfers, and shorter onboarding cycles often matter more than having one extra analytics widget. In a value-based procurement model, integration depth should rank above feature count.
Regulatory alignment, cybersecurity, and audit readiness should be compared early
Healthcare organizations increasingly need software that can withstand regulatory review, not just daily clinical use. Diagnostic imaging software should therefore be compared for documentation quality, change management, validation approach, and cybersecurity controls as early as the shortlisting stage. Waiting until contract negotiation often exposes gaps that delay procurement by 30–90 days.
For organizations operating in or supplying to regulated markets, alignment with MDR or IVDR-related expectations may affect how software functions are documented, updated, and risk-assessed. Even when the software itself is one part of a larger workflow, buyers should review intended use clarity, software lifecycle management, and evidence that updates do not undermine validated workflows.
Cybersecurity belongs in the same conversation. Imaging platforms are connected systems with user accounts, network interfaces, and stored patient data. At minimum, procurement teams should ask about encryption, patching cadence, access controls, log retention, and backup recovery objectives. A credible vendor or platform operator should be able to describe patch cycles such as monthly, quarterly, or risk-triggered releases, not respond in vague terms.
Audit readiness also matters for laboratory architects and enterprise buyers. If change logs, user permissions, system events, and validation records cannot be retrieved in a structured way, the software may create operational friction later. A practical benchmark is whether common audit evidence can be exported within 24–48 hours rather than assembled manually over several days.
H4-level review points for compliance screening
Documentation package
Check whether the software includes installation records, validation guidance, release notes, risk-management references, and clear revision history. These items are often more useful in procurement than broad marketing summaries.
Security operations
Confirm role-based access control, password policy support, session timeout options, and incident response commitments. A target response window of 4–24 hours for critical issues is a practical benchmark in many healthcare IT environments.
Validation and change control
Ask how updates are tested, how often major releases occur, and whether regression checks are available for connected systems. Quarterly review cycles are common, but the key question is whether workflow-impacting changes are visible before deployment.
- Require a documented software update and rollback process.
- Review whether logs support user actions, access events, and configuration changes.
- Check data retention settings against organizational policy, often 12 months or longer for operational logs.
- Confirm backup and restore testing frequency, ideally at least annually and after major infrastructure changes.
When these controls are reviewed early, procurement teams reduce the chance of selecting a system that is clinically useful but operationally difficult to govern. In imaging environments, compliance discipline is part of performance, not a separate administrative layer.
Measure total cost over the full software lifecycle, not only license price
One of the most common procurement mistakes is comparing diagnostic imaging software on upfront price alone. The better comparison is total cost over 3–5 years or, for larger networks, 5–7 years. This includes licenses, implementation, interfaces, storage, cybersecurity maintenance, training, workflow redesign, validation work, and support. A lower initial quote can become the more expensive choice if integration or archive costs escalate after go-live.
Operational teams should also estimate the cost of inefficiency. If each radiologist loses 20 minutes per day due to slow loading, fragmented navigation, or duplicate clicks, the annual productivity impact can exceed the apparent savings of a cheaper platform. Likewise, if operators need extra manual reconciliation for 2%–5% of studies, that creates recurring hidden labor cost.
Medical technology cost analysis should therefore combine direct and indirect factors. Direct factors include subscription or perpetual licensing, server or cloud costs, and vendor support. Indirect factors include downtime risk, re-training after poor usability, quality incident management, and future migration complexity if the platform cannot scale.
A procurement model grounded in technical benchmarking helps here. VSM-style evaluation emphasizes measurable parameters: implementation hours, interface count, archive growth rate, expected refresh intervals, and support SLAs. These factors turn software selection from a subjective preference into a more controlled engineering decision.
Lifecycle cost categories to compare
The following table is useful during vendor review meetings or internal business-case preparation because it frames price against real operational impact.
The key conclusion is simple: a good imaging platform is not necessarily the one with the lowest first-year spend. It is the one that delivers stable clinical performance, manageable compliance effort, and sustainable operating cost over the full service life.
Implementation, user adoption, and common buying mistakes
Even strong diagnostic imaging software can underperform if implementation is rushed. A practical rollout usually includes 3 phases: technical preparation, controlled validation, and monitored go-live. Depending on organizational size, each phase may take 1–3 weeks for a focused deployment or longer for multi-site programs. Skipping validation to save time often leads to months of user complaints and expensive post-launch corrections.
User adoption should be planned by role. Operators need workflow clarity, radiologists need performance consistency, IT teams need supportability, and decision-makers need measurable outcomes. The most effective projects define 5–7 acceptance criteria before go-live, such as study loading time, report turnaround consistency, integration stability, user access accuracy, and backup recovery readiness.
A frequent mistake is over-prioritizing AI add-ons or advanced visualization while neglecting base workflow. Another is failing to compare service support in detail. If the vendor or implementation partner cannot define training scope, escalation paths, or validation ownership, the organization inherits operational ambiguity from day one. That risk is especially high in regulated healthcare environments.
Independent benchmarking can reduce these mistakes. By translating engineering parameters into comparable evidence, organizations can distinguish between software that demos well and software that performs reliably in live care settings. This is particularly relevant for procurement directors, MedTech startups, and laboratory architects who must defend decisions beyond the initial purchase stage.
A practical 5-step evaluation path
- Define the clinical and operational use case, including modality mix, user roles, and expected daily study volume.
- Test image integrity, viewer performance, and workflow latency using representative cases rather than simplified demo files.
- Review interoperability, data governance, and healthcare compliance solutions with IT and compliance stakeholders together.
- Build a 3–5 year cost model that includes support, storage, validation, and upgrade effort.
- Run a limited pilot with measurable acceptance criteria before full deployment.
FAQ for buyers and operators
How many systems should be compared in a shortlist?
In many B2B healthcare procurements, 3–5 options are enough. Fewer than 3 may narrow perspective too early, while more than 5 often slows evaluation without improving decision quality.
What is a reasonable pilot duration?
For a single-site workflow, 2–4 weeks is usually enough to observe loading performance, user behavior, and integration stability. Larger, multi-site pilots may need 6–8 weeks.
What should procurement ask about support?
Ask for service hours, response times by severity, patch frequency, named ownership for interfaces, and escalation routes. These details are often more predictive of long-term satisfaction than feature counts.
Is cloud deployment always better?
Not always. Cloud can improve scalability and remote access, but the better option depends on network reliability, archive design, data governance, and integration requirements. The right choice should be validated case by case.
For organizations evaluating diagnostic imaging software today, the best first comparison points are clinical accuracy, interoperability, regulatory readiness, and lifecycle cost. These four dimensions shape safety, efficiency, and long-term return far more than surface-level feature lists. VSM supports this evidence-led approach by helping healthcare decision-makers examine technical integrity, compliance alignment, and operational reliability with greater precision.
If your team is planning a software upgrade, a new imaging workflow, or a procurement review across medical technology suppliers, now is the right time to benchmark before you buy. Contact VitalSync Metrics to discuss a structured evaluation, request a tailored comparison framework, or explore more healthcare-focused technical benchmarking solutions.
- EMS
- ESS
- EdTech
- procurement
- cybersecurity
- AR
- benchmarking
- healthcare procurement
- value-based procurement
- medical technology
- technical integrity
- medical equipment
- technical benchmarking
- data-driven
- clinical performance
- healthcare compliance
- Diagnostic Imaging
- medical technology supplier
- medical technology cost
- Diagnostic Imaging software
- medical equipment calibration
- medical equipment safety standards
- healthcare compliance solutions
Recommended News
- 2026.04.24Medical technology cost: where budgets go wrongDr. Alistair Thorne (Senior Biomedical Engineer)
- 2026.04.24Packaging choices that protect sterile instrumentsDr. Julian Rossi (RehabTech Specialist)
- 2026.04.24Mobility assist devices: which option fits bestSarah Jenkins (Laboratory Infrastructure Consultant)
The VitalSync Intelligence Brief
Receive daily deep-dives into MedTech innovations and regulatory shifts.
