Healthcare compliance solutions worth adopting now
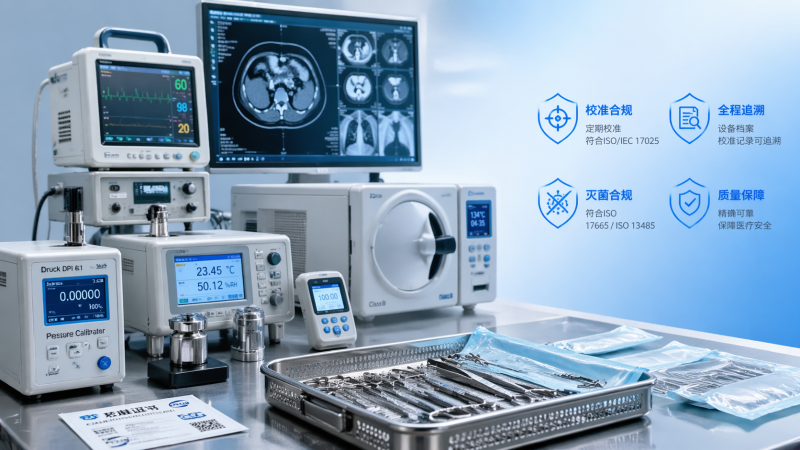
In a healthcare market shaped by stricter medical equipment safety standards, rising medical technology cost, and rapid medical technology advancements, adopting the right healthcare compliance solutions is now a strategic necessity. From wearable healthcare technology and Diagnostic Imaging software to surgical instruments sterilization and medical equipment calibration, decision-makers need verifiable, future-ready systems that reduce risk, support procurement confidence, and strengthen long-term operational performance.
For researchers, operators, procurement teams, and business leaders, compliance is no longer a narrow regulatory task handled at the end of a project. It now affects supplier qualification, total cost of ownership, implementation speed, maintenance planning, and cross-border commercialization. In practice, one weak link—such as incomplete calibration records, poor traceability, or weak MDR/IVDR documentation—can delay evaluation by 2–8 weeks and create avoidable downstream cost.
That is why healthcare organizations are shifting from passive document collection to structured compliance solutions that combine technical benchmarking, validation workflows, risk controls, and lifecycle monitoring. For organizations sourcing advanced medical technologies, the goal is not just to “meet requirements,” but to prove performance, maintain audit readiness, and protect long-term clinical reliability.
Why healthcare compliance solutions now influence procurement and operational strategy
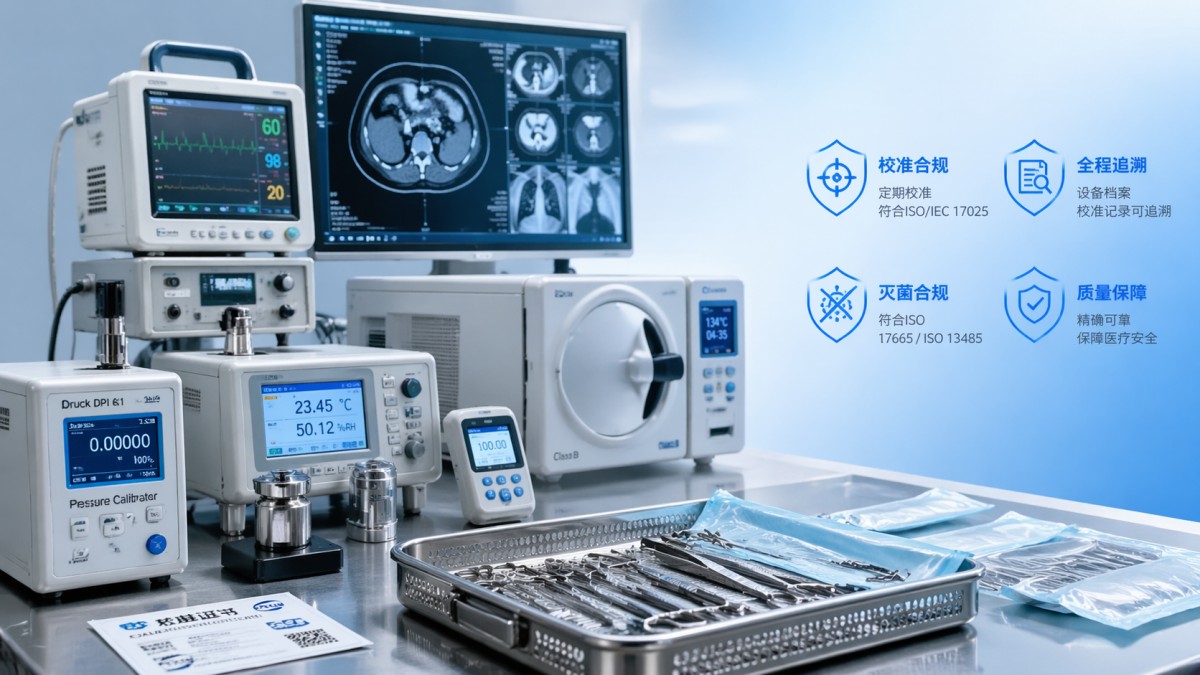
Healthcare compliance solutions now sit at the intersection of regulation, engineering verification, and purchasing discipline. Hospitals and laboratories are under pressure to make better decisions with tighter budgets, while MedTech startups must show that device claims are supported by measurable performance. In both cases, compliance has become a decision framework rather than a paperwork exercise.
In value-based procurement environments, technical integrity matters as much as price. A lower-cost device may appear attractive, but if it requires calibration every 30 days instead of every 90–180 days, or if it lacks clean validation evidence for software updates, the lifetime operating burden can rise sharply. Procurement teams increasingly evaluate 4 core areas: regulatory status, performance consistency, serviceability, and evidence quality.
This shift is especially visible in wearable healthcare technology, diagnostic platforms, sterilization workflows, and imaging systems. These categories involve multiple risk layers: sensor drift, software version control, contamination risk, component fatigue, and interoperability gaps. Compliance solutions help convert those risks into clear checklists, test thresholds, and approval gates that non-engineering stakeholders can also understand.
What decision-makers are really trying to avoid
The most common procurement concern is not simply nonconformity. It is hidden uncertainty. A device may pass initial demonstrations but later fail under repeated use, transport stress, or real-world cleaning cycles. Compliance programs reduce that uncertainty by connecting documentation with actual performance evidence, such as repeatability bands, sterilization cycle tolerance, or signal stability under defined environmental conditions.
The table below outlines how compliance affects key buying and operating decisions across common healthcare technology categories.
The pattern is clear: compliance weaknesses rarely remain isolated. They spread into maintenance load, downtime, procurement disputes, and quality risk. For that reason, many buyers now ask for benchmark-backed whitepapers, defined test methods, and lifecycle data instead of relying only on marketing brochures.
Core healthcare compliance solutions worth adopting now
Not every organization needs the same compliance stack, but several solution categories are becoming essential across hospitals, laboratories, and MedTech supply chains. The strongest approach combines documentation readiness with engineering verification and post-installation control. This is particularly important where performance degradation may appear gradually over 3–12 months rather than during acceptance testing.
1. Technical benchmarking and independent verification
Independent benchmarking helps buyers compare products using standardized measurements instead of inconsistent vendor claims. For example, wearable sensor evaluation may include signal-to-noise thresholds, repeatability across 50–200 cycles, and response consistency under temperature ranges such as 10°C–35°C. This creates a more objective starting point for sourcing decisions.
2. Digital document control and traceability systems
A digital compliance system should track specifications, revisions, test records, maintenance logs, and approval history in one controlled environment. Teams that still rely on scattered spreadsheets and email attachments often struggle during audits or supplier reviews. A unified platform can shorten document retrieval from several hours to a few minutes while reducing version confusion.
3. Calibration and validation scheduling
For medical equipment calibration, interval planning is as important as the calibration event itself. Critical devices may require checks every 6 months, while lower-risk instruments may follow 12-month intervals if performance history is stable. Good compliance solutions support interval adjustment based on usage intensity, deviation trends, and risk classification.
4. Sterilization compliance monitoring
For surgical instruments sterilization, compliance should cover process validation, packaging integrity, load configuration, and material durability. Repeated exposure to heat, chemicals, or moisture can change surface properties over time. A robust monitoring program reviews not only pass/fail outcomes, but also cycle consistency over 20, 50, or 100 runs where relevant.
Recommended adoption priorities
- Start with high-risk, high-utilization assets that directly affect diagnosis, intervention, or patient monitoring.
- Prioritize systems with recurring software updates, measurable drift risk, or heavy cleaning and sterilization exposure.
- Use a 3-step rollout: baseline assessment, gap closure, and ongoing monitoring with quarterly review points.
- Define at least 5 mandatory record types: specification, validation result, calibration evidence, maintenance history, and deviation log.
Organizations that adopt these healthcare compliance solutions early often gain a practical advantage: faster supplier comparison, stronger audit readiness, and better alignment between technical teams and procurement leaders. The key is to choose solutions that turn raw data into decision-ready evidence.
How to evaluate healthcare compliance solutions before purchase
Selecting a compliance solution requires more than checking whether a vendor understands regulatory language. Buyers should evaluate whether the solution can support real operating conditions, multi-site documentation, and measurable performance review. A system that looks comprehensive on paper may still fail if it cannot integrate with maintenance workflows, acceptance testing, or supplier qualification processes.
A useful evaluation model includes 6 criteria: regulatory coverage, evidence depth, implementation effort, interoperability, audit usability, and lifecycle cost. For hospital groups and laboratory planners, interoperability is especially important because isolated compliance tools often create extra manual work instead of reducing it.
Practical comparison points for procurement teams
Before issuing a purchase order, request concrete examples of how the provider handles deviations, specification changes, and recurring validation. Ask whether the platform supports revision tracking, approval timestamps, and evidence export for external review. A response time standard of 24–72 hours for critical document support is often a reasonable baseline in B2B healthcare environments.
The table below can be used as a working checklist during supplier evaluation or internal approval meetings.
A good solution should also fit the user profile. Operators need simple workflows and clear alerts. Researchers need access to raw performance evidence. Procurement teams need comparison-ready outputs. Executives need risk visibility and investment justification. If one of these groups is ignored, adoption usually weakens within the first 90 days.
Common purchasing mistakes
- Choosing a compliance tool based only on document storage without testing or benchmark capability.
- Ignoring calibration and sterilization workflows during software selection.
- Accepting supplier declarations without asking for defined methods, tolerances, or repeatability evidence.
- Underestimating training needs for frontline operators who handle the system daily.
These issues are avoidable when compliance selection is treated as an operational design decision rather than a narrow quality department task.
Implementation roadmap: from compliance gap to controlled healthcare operation
Even the best healthcare compliance solutions fail if implementation is rushed or fragmented. A practical rollout should move in stages, with clear ownership and measurable checkpoints. For most organizations, a 4-stage approach works well: assessment, prioritization, deployment, and performance review. Depending on site complexity, this may take 4–12 weeks for the first wave.
Stage 1: Map devices, processes, and risk points
Begin by identifying all assets and workflows that require compliance control. This should include medical equipment calibration schedules, software validation needs, sterilization dependencies, and any outsourced testing activities. A structured inventory often reveals 10%–20% of devices with unclear ownership or outdated records.
Stage 2: Define acceptance criteria and evidence requirements
Every critical process should have predefined acceptance thresholds. For example, a device may require calibration deviation within a specified tolerance band, or a software release may require traceable validation before activation. Without agreed criteria, teams tend to approve based on familiarity or urgency instead of evidence.
Stage 3: Train users and assign escalation paths
Training should be role-based. Operators need workflow clarity, procurement teams need document interpretation guidance, and technical reviewers need deeper benchmark understanding. A short training cycle of 2–3 sessions is often enough for basic adoption, but high-risk functions may require competency checks and refreshers every 6–12 months.
Stage 4: Review drift, deviations, and supplier performance
Compliance is dynamic. Once the system is live, teams should review recurring exceptions, overdue records, calibration deviations, and supplier responsiveness. Quarterly review is a practical baseline, while high-risk categories may need monthly review until stability is proven. This turns compliance into a management tool rather than a static archive.
Implementation checklist
- Assign 1 owner for each device group, software module, or sterilization process.
- Define 3 document states at minimum: draft, approved, and obsolete.
- Set alert rules for calibration due dates, expired validation, and unresolved deviations.
- Review supplier evidence packages before first use, not after deployment.
- Track corrective actions with closure targets such as 7, 14, or 30 days based on severity.
Organizations that follow a phased rollout usually gain faster internal adoption because users understand how compliance supports daily work. The biggest benefit is not only audit readiness, but more consistent equipment performance and fewer avoidable interruptions.
FAQ: questions buyers and healthcare operators ask most often
How do I know which healthcare compliance solutions should be implemented first?
Start with systems linked to patient monitoring, diagnosis, sterile processing, or critical measurement. If a device failure, drift event, or documentation gap could disrupt care or delay procurement approval, it should be in the first wave. A practical rule is to rank assets by risk, usage frequency, and replacement cost, then prioritize the top 20% that carry the highest operational impact.
What should procurement teams request from suppliers besides certificates?
Ask for benchmark reports, test methods, performance tolerances, maintenance intervals, software change procedures, and document traceability examples. For medical equipment calibration, request interval rationale and deviation history if available. For Diagnostic Imaging software, ask how updates are validated and whether rollback controls exist.
How long does implementation usually take?
For a focused rollout covering one site or one technology category, 2–6 weeks is common. A broader deployment across multiple departments may take 8–12 weeks, especially if historical records must be cleaned or migrated. The timeline depends less on software installation and more on evidence review, user training, and process alignment.
Are compliance solutions only for large hospital groups?
No. Smaller clinics, diagnostic labs, and MedTech startups often gain significant value because they have less room for documentation gaps or rework. A lighter system with clear controls, essential traceability, and defined review cycles can still reduce risk and improve sourcing decisions without creating unnecessary administrative burden.
What role does independent benchmarking play?
Independent benchmarking helps separate marketing claims from technical reality. It is particularly useful when comparing competing products with similar stated specifications. By testing measurable factors such as durability, repeatability, signal quality, or fatigue limits, buyers can make decisions based on evidence instead of assumptions.
Healthcare compliance solutions are now central to safer procurement, stronger validation, and more reliable operations. The organizations making the best decisions are those that connect regulatory readiness with measurable engineering evidence, practical workflows, and lifecycle visibility. That applies whether you are qualifying wearable healthcare technology, reviewing Diagnostic Imaging software, strengthening surgical instruments sterilization controls, or improving medical equipment calibration governance.
VitalSync Metrics (VSM) supports this shift by helping healthcare decision-makers evaluate technical integrity with greater precision and less promotional noise. If your team needs clearer benchmarking, stronger compliance documentation, or a more defensible sourcing framework, now is the time to move from fragmented checks to a structured solution strategy. Contact us to discuss your requirements, request a tailored evaluation approach, or learn more about compliance-focused benchmarking for your healthcare supply chain.
- EMS
- ESS
- surgical instruments
- PPE
- healthcare technology
- patient monitoring
- EdTech
- procurement
- cybersecurity
- AR
- supply chain
- Cement
- benchmarking
- value-based procurement
- MedTech supply chain
- medical technology
- technical integrity
- medical equipment
- surgical instruments
- MedTech supply
- technical benchmarking
- supply chain
- healthcare supply chain
- healthcare technology
- healthcare compliance
- Diagnostic Imaging
- Packaging
- medical technology cost
- Diagnostic Imaging software
- medical technology advancements
- medical equipment calibration
- surgical instruments sterilization
- medical equipment safety standards
- wearable healthcare technology
- healthcare compliance solutions
Recommended News
- 2026.04.24Medical technology cost: where budgets go wrongDr. Alistair Thorne (Senior Biomedical Engineer)
- 2026.04.24Packaging choices that protect sterile instrumentsDr. Julian Rossi (RehabTech Specialist)
- 2026.04.24Mobility assist devices: which option fits bestSarah Jenkins (Laboratory Infrastructure Consultant)
The VitalSync Intelligence Brief
Receive daily deep-dives into MedTech innovations and regulatory shifts.
