How to choose surgical instruments sterilization methods
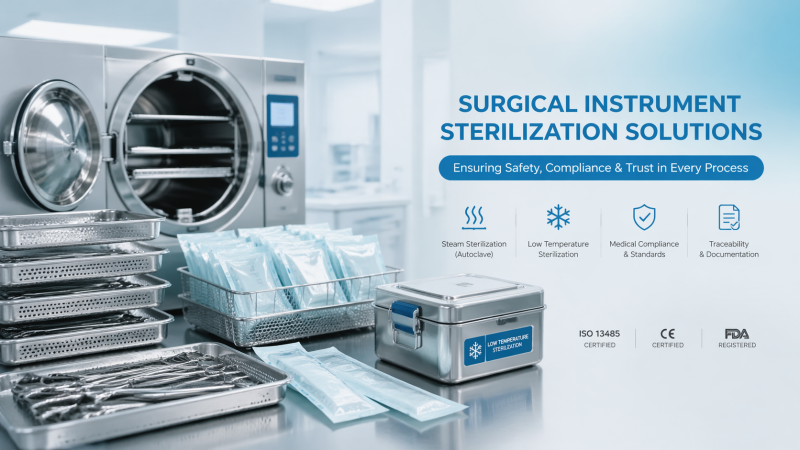
Choosing the right surgical instruments sterilization method is critical for patient safety, workflow efficiency, and healthcare compliance solutions. From steam and low-temperature systems to medical equipment sterilization protocols, each option affects instrument lifespan, medical equipment safety standards, and long-term cost control. This guide helps researchers, operators, procurement teams, and decision-makers evaluate practical factors behind safer, more reliable sterilization choices.
How should healthcare teams compare surgical instruments sterilization methods?
A useful comparison starts with one basic truth: no single surgical instruments sterilization method fits every instrument, department, and procurement model. In most hospitals, laboratories, and ambulatory settings, the decision involves at least 4 core variables: material compatibility, device complexity, processing time, and documentation requirements. A method that works well for stainless steel trays may damage heat-sensitive polymers, adhesives, optics, or powered components.
For operators, the first concern is consistent microbial reduction without workflow disruption. For procurement teams, the priority often shifts to total cost of ownership over 3–7 years, including consumables, maintenance, chamber capacity, and instrument replacement risk. For decision-makers, the bigger issue is whether the sterilization route aligns with medical equipment safety standards, internal validation practice, and cross-border compliance expectations.
In practice, the most common choices include steam sterilization, hydrogen peroxide vapor or plasma systems, and ethylene oxide for selected applications. Dry heat exists, but its use is more limited because cycle durations can be longer and material suitability narrower. The right evaluation is therefore not only technical. It must connect reprocessing science, procurement logic, and operational uptime.
VitalSync Metrics (VSM) approaches this evaluation from an engineering and benchmarking perspective. Instead of accepting supplier claims at face value, buyers should compare chamber performance, cycle repeatability, load configuration limits, material compatibility statements, and traceability outputs. That is especially important in value-based procurement, where a lower acquisition price can still produce higher downstream cost if rework, instrument degradation, or nonconformance events increase.
A practical comparison framework
- Assess instrument categories first: rigid stainless steel, lumened instruments, microsurgical tools, polymer-containing sets, and powered or heat-sensitive devices often require different sterilization decisions.
- Review cycle constraints: a 20–60 minute turnaround may be acceptable in one department, while central sterile operations may plan around batch windows of several hours including drying, aeration, or release checks.
- Verify documentation expectations: many facilities now require digital logs, load records, cycle parameter export, and validation support for audits and quality reviews.
- Estimate hidden replacement costs: repeated exposure to incompatible conditions can shorten instrument service life long before the sterilizer itself reaches end of life.
The table below summarizes common sterilization options from a procurement and operating perspective. The values are directional rather than brand-specific, because actual cycles depend on load design, packaging, local protocol, and validated use instructions.
This comparison shows why sterilization choice must be linked to actual device families and validated instructions for use. Procurement teams that compare only cycle speed often miss the operational consequences of packaging restrictions, lumen claims, wet load risks, and consumable dependency. A balanced review is more defensible in quality meetings and more reliable over the full service life of the instrument fleet.
Which sterilization method fits specific instrument and workflow scenarios?
Application fit matters more than generic popularity. Steam remains the default choice in many hospitals because it is widely validated for reusable metal instruments, economical at scale, and practical for high-throughput central sterile departments. However, once a tray includes camera heads, polymer seals, long narrow lumens, or manufacturer-restricted components, medical equipment sterilization protocols need a more selective path.
For operating staff, the question is often turnaround. A department handling 10–20 instrument sets per shift needs predictable release timing. If the sterilization method introduces long hold periods, packaging failures, or repeat cycles, surgical scheduling may be affected. For infection prevention teams, the question is narrower: can the selected method reach the most difficult areas of the device consistently under validated loading conditions?
Research facilities and prototype labs add another layer. They may process small batches, mixed materials, or nonstandard assemblies. In such cases, a low-temperature route may protect sensitive components, but only if compatibility data, residue considerations, and process validation are clear. A method suitable for 5 trays per day may be uneconomical for 100 trays per day.
VSM typically recommends mapping sterilization decisions by 3 dimensions: device design risk, throughput need, and compliance burden. That converts an abstract technical debate into a sourcing and workflow model that procurement, operators, and quality managers can all evaluate on the same page.
Scenario-based selection checkpoints
Routine metal instrument sets
When trays are primarily stainless steel and validated for moist heat, steam sterilization is usually the most practical option. It supports batch throughput, commonly available infrastructure, and comparatively straightforward operator training. The main control points are proper cleaning, packaging integrity, load configuration, and complete drying before release.
Heat-sensitive and mixed-material devices
If devices contain sensitive plastics, electronics, optics, or bonded components, low-temperature sterilization often becomes the preferred route. Even then, teams must confirm lumen compatibility, material-specific restrictions, and packaging requirements. Shorter low-temperature cycles can look attractive, but they do not eliminate the need for process controls and staff discipline.
Complex devices with narrow channels
Complex designs demand caution. A device with long or narrow internal channels may be technically sterilizable only under specific loading conditions defined by the manufacturer. This is where procurement decisions often fail: teams purchase a sterilizer first, then discover that certain high-value instruments require different accessories, restricted loads, or a separate method entirely.
The next table is useful when departments need to connect application scenarios with method selection, equipment planning, and risk awareness.
The key insight is that method selection is often portfolio-based rather than single-system based. A facility may standardize steam for 70%–90% of routine loads while retaining a low-temperature pathway for selected devices. That hybrid logic can reduce both overinvestment and instrument damage, especially when driven by actual use patterns rather than vendor preference.
What should procurement teams and decision-makers verify before buying?
Purchasing a sterilization solution is not the same as purchasing a chamber. The buying decision should cover at least 5 check areas: validated cycle capability, instrument compatibility, digital traceability, service model, and long-term operating cost. Many procurement mistakes happen because teams compare list price but do not compare downtime exposure, consumable demand, staff retraining needs, or restrictions on future device portfolios.
Operators should ask whether the method supports realistic load sizes and daily cadence. A sterilizer may perform well in ideal demonstrations but struggle under mixed packaging, irregular trays, or peak-day pressure. Decision-makers should ask a different question: what happens over the next 24–60 months if the hospital adds more complex minimally invasive devices, outsourced service obligations, or stricter documentation rules?
This is where VSM’s independent benchmarking position is valuable. In the MedTech and Life Sciences supply chain, procurement risk increasingly comes from the gap between brochure claims and engineering-grade performance. A credible evaluation should review cycle data outputs, preventive maintenance assumptions, utility dependence, replacement parts exposure, and whether the sterilization platform can support a standardized whitepaper-style comparison across sites or tenders.
For organizations operating under value-based procurement rules, the best choice is often the option with the most predictable total sterilization performance, not merely the fastest headline cycle. A slower but more stable process can outperform a nominally faster system if it reduces load recalls, instrument wear, repeat processing, and operator workarounds.
A procurement checklist for sterilization method selection
- Confirm the exact instrument mix for the next 12–24 months, not only the current inventory. Procurement should include upcoming specialty devices, trial programs, and replacements.
- Request method-specific compatibility evidence for heat-sensitive items, lumened devices, polymer components, and accessories. General marketing claims are not enough.
- Review the full operating model: consumables, utilities, maintenance windows, software logging, alarm handling, and staff competency requirements.
- Define 3–5 acceptance criteria before evaluation, such as throughput range, traceability capability, audit readiness, and service response expectations.
- Model worst-case workflow impact. Include repeat cycle probability, downtime scenarios, and whether a backup sterilization path exists.
Where hidden cost usually appears
Hidden cost is rarely one large expense. It usually appears in small repeated losses: extra packaging, missed release windows, more frequent instrument refurbishment, unplanned service visits, consumable waste, and staff time spent resolving cycle exceptions. Over a 3-year or 5-year review period, these factors can reshape the economics of the project far more than the initial quotation.
A sound purchasing decision therefore requires more than a price sheet. It needs a technical review process that can translate sterilization performance into procurement confidence. That is the value of independent comparison logic: it helps buyers identify whether a solution is genuinely fit for intended use, or merely attractive in sales presentation.
How do standards, validation, and compliance influence sterilization method choice?
Sterilization is never just a technical utility function. It is part of a larger quality and compliance system. Healthcare organizations and MedTech suppliers must align method choice with validated instructions for use, documented reprocessing workflows, and applicable standards or regulatory expectations. In cross-border procurement, the burden is often higher because technical files, supplier documentation, and process traceability may be reviewed by multiple internal and external stakeholders.
For that reason, sterilization method choice should be documented around 4 layers: cleaning prerequisite, sterilization process parameters, packaging and load definition, and release evidence. When instruments are introduced into regulated markets or linked to MDR/IVDR-related supply chain expectations, decision-makers should also check whether supporting technical documentation is complete, current, and usable for tender review or quality assessment.
Medical equipment safety standards are especially relevant when sterilization systems interact with digital tracking, validation records, and service documentation. The stronger the documentation chain, the easier it becomes to investigate deviations, train operators, and defend procurement choices. Weak documentation may not fail immediately, but it raises risk during audits, incident reviews, and contract renewals.
VSM’s role in this environment is not to replace regulatory interpretation, but to support technical clarity. By converting manufacturing and performance parameters into standardized benchmarking views, organizations can compare solutions with less ambiguity and more engineering discipline. That is particularly useful when multiple stakeholders need one common evidence base before approval.
Compliance questions worth asking early
- Does the sterilization method align with the instrument manufacturer’s validated reprocessing instructions, including load limits and accessory use?
- Are cycle records exportable, reviewable, and retained according to internal quality system expectations?
- Can the supplier explain maintenance, calibration, and validation support without relying on vague language?
- If the organization operates internationally, are documentation formats suitable for tenders, technical review, and supplier qualification across regions?
Why documentation quality affects procurement confidence
In many projects, two systems may appear comparable on the surface, yet differ significantly in documentation maturity. One provides clear process boundaries, service intervals, data outputs, and compatibility statements. The other offers broad claims but weak usable detail. The first option usually creates lower long-term risk, even if the acquisition cost is higher. This is a critical issue for enterprise decision-makers who must justify procurement with evidence rather than assumptions.
What common mistakes should users avoid, and what questions do buyers ask most often?
Several mistakes repeat across healthcare settings. Teams often assume that if a sterilization method is widely used, it must be suitable for all instruments. They also confuse disinfection with sterilization, underestimate packaging and drying variables, or ignore the cumulative effect of repeated cycles on instrument condition. In busy departments, these assumptions can persist for months before they appear as workflow disruption, repair cost, or compliance findings.
Another common error is treating procurement, operation, and quality assurance as separate conversations. In reality, the method should be selected through one integrated review. If users prioritize speed, procurement prioritizes price, and quality prioritizes documentation without a shared framework, the final choice may satisfy nobody. This is why structured evaluation workshops, even over 2–3 review rounds, often produce better outcomes than one-time vendor comparisons.
Below are frequently asked questions that reflect real search intent from information researchers, operators, buyers, and executive teams. The answers focus on practical decision points rather than generic descriptions.
How do I choose between steam and low-temperature sterilization?
Start with device compatibility, not preference. If the instrument is robust, metallic, and validated for moist heat, steam is often the first choice because it supports efficient batch processing and broader routine use. If the device contains heat-sensitive materials, optics, electronics, or restrictive manufacturer instructions, low-temperature sterilization may be necessary. The deciding factors are validated compatibility, workflow fit, and full operating cost.
Which sterilization method is best for delicate surgical instruments?
There is no universal best option. Delicate instruments often benefit from low-temperature systems, but only when the exact material set, channel design, and packaging configuration are supported. Some delicate instruments can still tolerate steam under specific conditions. The correct answer should always come from validated reprocessing instructions plus a practical review of turnover time and traceability requirements.
What should procurement teams ask suppliers before final selection?
Ask for 5 concrete items: compatible device categories, cycle documentation outputs, routine maintenance assumptions, consumable dependency, and service response model. Also ask what happens under non-ideal conditions such as mixed loads, interrupted cycles, or accessory shortages. These questions reveal whether the supplier understands real sterile processing operations or only ideal demonstration conditions.
How long does implementation usually take?
Implementation timing varies by facility size and method complexity. A simple replacement project may move through review, installation, qualification, and training in a few weeks, while a more complex multi-stakeholder rollout can take 4–12 weeks or longer. The longest delays usually come from documentation review, site utilities, validation planning, and alignment with existing medical equipment sterilization protocols.
Can one facility rely on a single sterilization method?
Sometimes yes, often no. Facilities with standardized metal instrument inventories may operate successfully with a mostly steam-based model. But hospitals that support minimally invasive surgery, specialty devices, mixed-material components, or external service demands frequently need a dual-method strategy. The goal is not to minimize equipment count at any cost; it is to minimize operational and compliance risk while preserving instrument integrity.
Why work with an independent benchmarking partner before finalizing a sterilization strategy?
In healthcare procurement, uncertainty is expensive. The challenge is rarely finding suppliers. The challenge is verifying whether the proposed sterilization method will hold up under real operating conditions, support medical equipment safety standards, and remain defensible during audits, scaling, or cross-site rollout. An independent benchmarking approach reduces that uncertainty by replacing broad claims with structured technical comparison.
VitalSync Metrics (VSM) supports buyers, operators, and decision-makers who need more than brochure-level information. We help translate technical parameters into procurement-ready evaluation logic, including compatibility review, workflow fit, compliance documentation readiness, and long-term reliability questions. That is particularly valuable for hospital procurement leaders, MedTech startups, and laboratory planners working in increasingly regulated, digitally integrated supply chains.
If you are reviewing surgical instruments sterilization methods, we can help you clarify 6 practical decision areas: method suitability by device type, cycle and throughput expectations, compliance documentation depth, service and maintenance assumptions, integration with existing reprocessing workflows, and total cost implications over the expected operating period. This makes internal approval faster and vendor comparisons more objective.
Contact VSM if you need support with parameter confirmation, sterilization method selection, delivery planning, custom evaluation frameworks, documentation review for MDR/IVDR-sensitive supply chains, sample-based technical discussions, or quotation-stage comparison. A structured assessment now can prevent costly mismatches later in procurement, implementation, and daily clinical use.
- EMS
- ESS
- surgical instruments
- PPE
- life sciences
- EdTech
- procurement
- AR
- supply chain
- Cement
- benchmarking
- healthcare procurement
- value-based procurement
- medical equipment
- surgical instruments
- hospital procurement
- life sciences
- supply chain
- long-term reliability
- healthcare compliance
- Sterilization Systems
- Packaging
- medical equipment sterilization
- surgical instruments sterilization
- medical equipment safety standards
- healthcare compliance solutions
Recommended News
- 2026.04.24Medical technology cost: where budgets go wrongDr. Alistair Thorne (Senior Biomedical Engineer)
- 2026.04.24Packaging choices that protect sterile instrumentsDr. Julian Rossi (RehabTech Specialist)
- 2026.04.24Mobility assist devices: which option fits bestSarah Jenkins (Laboratory Infrastructure Consultant)
The VitalSync Intelligence Brief
Receive daily deep-dives into MedTech innovations and regulatory shifts.
