lndustry Portal
Global B2B Solutions
 LFP battery safety for medical devices is not just a thermal issueLFP battery safety for medical devices goes beyond heat—see how EMC testing for medical electronics and ISO 13485 audit requirements reveal true clinical reliability.Apr 16, 2026Sarah Jenkins (Laboratory Infrastructure Consultant)
LFP battery safety for medical devices is not just a thermal issueLFP battery safety for medical devices goes beyond heat—see how EMC testing for medical electronics and ISO 13485 audit requirements reveal true clinical reliability.Apr 16, 2026Sarah Jenkins (Laboratory Infrastructure Consultant) Medical grade plastic certificates do not always prove biocompatibilityMedical grade plastic certificates do not guarantee compliance with biocompatibility testing standards. Learn how to verify real device safety, reduce sourcing risk, and strengthen regulatory decisions.Apr 16, 2026Dr. Julian Rossi (RehabTech Specialist)
Medical grade plastic certificates do not always prove biocompatibilityMedical grade plastic certificates do not guarantee compliance with biocompatibility testing standards. Learn how to verify real device safety, reduce sourcing risk, and strengthen regulatory decisions.Apr 16, 2026Dr. Julian Rossi (RehabTech Specialist) Autoclave sterilization validation for porous loads needs extra proofAutoclave sterilization validation for porous loads needs extra proof—learn how to verify penetration, dryness, and repeatability while aligning with ISO 13485 audit requirements and FDA MDR compliance checklist.Apr 16, 2026Dr. Alistair Thorne (Senior Biomedical Engineer)
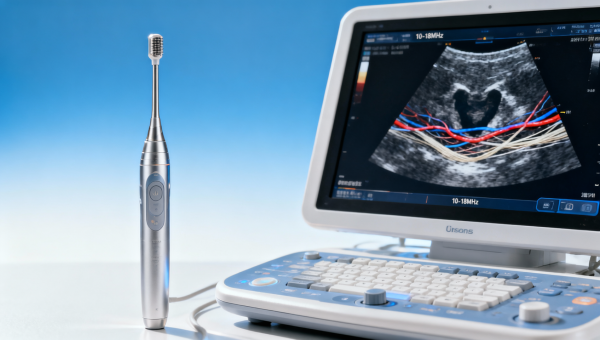
Autoclave sterilization validation for porous loads needs extra proofAutoclave sterilization validation for porous loads needs extra proof—learn how to verify penetration, dryness, and repeatability while aligning with ISO 13485 audit requirements and FDA MDR compliance checklist.Apr 16, 2026Dr. Alistair Thorne (Senior Biomedical Engineer) Choosing ultrasound transducer frequency range for shallow structuresUltrasound transducer frequency range for shallow structures explained with buyer-focused benchmarks, ISO 13485 audit requirements, and signal to noise ratio insights for smarter procurement.Apr 16, 2026Lydia Vancini (Regulatory Compliance Lead)
Choosing ultrasound transducer frequency range for shallow structuresUltrasound transducer frequency range for shallow structures explained with buyer-focused benchmarks, ISO 13485 audit requirements, and signal to noise ratio insights for smarter procurement.Apr 16, 2026Lydia Vancini (Regulatory Compliance Lead) EMC testing for medical electronics before redesign saves hidden costsEMC testing for medical electronics reveals design risks early, reduces redesign costs, and supports FDA MDR compliance checklist readiness for safer, more reliable device launches.Apr 16, 2026Lydia Vancini (Regulatory Compliance Lead)
EMC testing for medical electronics before redesign saves hidden costsEMC testing for medical electronics reveals design risks early, reduces redesign costs, and supports FDA MDR compliance checklist readiness for safer, more reliable device launches.Apr 16, 2026Lydia Vancini (Regulatory Compliance Lead) Patient monitor PCBA manufacturer checks that reduce field failuresPatient monitor PCBA manufacturer checks that cut field failures: learn how emc testing for medical electronics and signal to noise ratio in patient monitors improve reliability before deployment.Apr 16, 2026Lydia Vancini (Regulatory Compliance Lead)
Patient monitor PCBA manufacturer checks that reduce field failuresPatient monitor PCBA manufacturer checks that cut field failures: learn how emc testing for medical electronics and signal to noise ratio in patient monitors improve reliability before deployment.Apr 16, 2026Lydia Vancini (Regulatory Compliance Lead) IVD analyzer distributor support matters most after installationIVD analyzer distributor support matters most after installation—discover how faster service, training, compliance records, and uptime protection reduce risk and improve long-term lab performance.Apr 16, 2026Dr. Hideo Tanaka (Imaging Systems Analyst)
IVD analyzer distributor support matters most after installationIVD analyzer distributor support matters most after installation—discover how faster service, training, compliance records, and uptime protection reduce risk and improve long-term lab performance.Apr 16, 2026Dr. Hideo Tanaka (Imaging Systems Analyst) Dental implant wholesale pricing changes once traceability is includedDental implant wholesale pricing now depends on traceability, biocompatibility testing standards, ISO 13485 audit requirements, and FDA MDR compliance checklist readiness—learn how to compare suppliers with confidence.Apr 16, 2026Dr. Alistair Thorne (Senior Biomedical Engineer)
Dental implant wholesale pricing changes once traceability is includedDental implant wholesale pricing now depends on traceability, biocompatibility testing standards, ISO 13485 audit requirements, and FDA MDR compliance checklist readiness—learn how to compare suppliers with confidence.Apr 16, 2026Dr. Alistair Thorne (Senior Biomedical Engineer) Surgical instrument kits OEM projects fail when tolerances stack upSurgical instrument kits OEM projects fail when tolerance stack-up is missed. Learn how ISO 13485 audit requirements, autoclave sterilization validation, and medical grade plastic certificates reveal hidden sourcing risks.Apr 16, 2026Dr. Alistair Thorne (Senior Biomedical Engineer)
Surgical instrument kits OEM projects fail when tolerances stack upSurgical instrument kits OEM projects fail when tolerance stack-up is missed. Learn how ISO 13485 audit requirements, autoclave sterilization validation, and medical grade plastic certificates reveal hidden sourcing risks.Apr 16, 2026Dr. Alistair Thorne (Senior Biomedical Engineer)
 {item.title}{item.description}{item.publish_time}{item.goods_category_sif3qfva9d}
{item.title}{item.description}{item.publish_time}{item.goods_category_sif3qfva9d}Search News
Popular Tags
Industry Overview
The global commercial kitchen equipment market is projected to reach $112 billion by 2027. Driven by urbanization, the rise of e-commerce food delivery, and strict hygiene regulations.